Basic Sciences
(Biochemistry, Plant physiology, Microbiology, Agricultural Chemistry)
Introduction
The Division of Basic Science is comprised of four disciplines Plant Physiology, Plant Biochemistry, Microbiology, and Agricultural chemicals. This Division is engaged in carrying out basic & strategic research on different aspects of pulses as per the mandate of the Institute. The Division is well equipped with modern facilities to carry out basic and strategic research viz., physiological constraints to productivity, improved adaptation to abiotic stresses, estimation of health-promoting bioactive substances, pesticide residue analysis, and improving phosphate solubilization and BNF through microbes in pulses.
Thrust Areas:
- Abiotic Stress PhysiologyEvaluation, phenotyping & physiological characterization of tolerance to major abiotic stresses.
- Production PhysiologyUnderstanding major yield barrier & stability mechanisms of pulses. Physiological mechanisms of high water & nutrient-use efficiency.
- Pulses for health and nutritional securityChemical profiling for nutritional and anti-nutritional bioactive components in pulses.
- Pesticide residue analysisDissipation study of herbicides in different environmental matrices
- Phosphate solubilization through microbes.Improving P-solubilization from rock-phosphate & enhancing P-use efficiency through PSB.
- Improving BNF through microbesEnhancing BNF through facilitating nutrients supply using microorganisms
- Improving water use efficiency through PGPRMitigating drought effect by plant growth-promoting bacteria
Achievements
Crop Physiology
- Physiological traits like RWC, SLW & DTI were found effective field screening tools for evaluating germplasm linesunder rainfed situation.
- Genotypes ICCV 10, BG 362, ICC 4958 had wide adaptation & appears to be high yielding genotypes under drought conditions.
- Genotypes PG 96006 & IPC 97-67 showed higher root mass even during podding& maturity stage expressing higher grain yield under drought condition.
Genotypic variation in root system

- An indigenous technology has been developed for root studies using the four tier pot system & under rainfed conditions the root length was measured up to 1.25 m length using this technique.
- Non-destructive methods were developed for large-scale screening of chickpea donors with high root system. Wooden structure with front glass windows filled with soil was made for constant monitoring of root development.
- Methodology for osmotic adjustment (OA) imparting tolerance to drought was perfected for screening large numbers of germplasm.
- Significant variability in leaflet size, shape, leaf length, number and canopy area was observed in chickpea. The leaflet size was found to have definite role in conserving moisture and plant-water status under drought.

- Plant types of chickpea for different agroclimatic zones have been identified which are best adapted to corresponding climates under which they are grown.


- Field screening for drought tolerance in chickpea based on drought susceptibility index (DSI) was found to be correlated with grain yield under drought
- Biomass was critical determinant of grain yield under drought.
- Indigenous techniques for root screening were developed based upon growing plants under PVC tubes and then cutting the tubes to expose roots intact through flow of water.
- Large genotypic variation in root traits was observed.
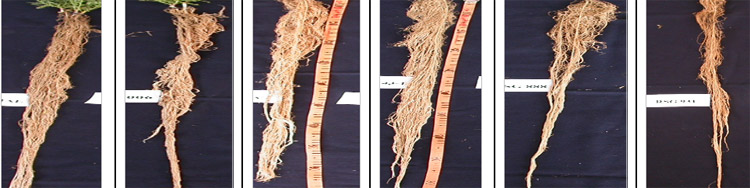
- Early flowering , quick biomass accumulation rate and high rooting depth and biomass were found to be the best strategies for adaptation of chickpea under drought
- ICC 4958, RSG 143-1, RSG 888, BGD 72, PG5, PG 96006, K 850, JG 14, JG 16, Vijay have been identified as drought tolerant chickpea genotypes/varieties.
Strategic adaptation of chickpea to high temperature
- Terminal heat stress during reproductive phase is a serious yield limiting constraint in chickpea causing.
- (i) Forced maturity
- (ii) Incomplete grain filling
- (iii) Hard and deformed seeds
- (iv)Faster maturity.
- A large number of chickpea collection from ICRISAT were evaluated for their thermo-tolerance and grain yield based upon Membrane injury level, Chlorophyll fluorescence Parameters (iv) pollen germination at high temperature > 350C and heat susceptibility index
- Contrasting heat tolerant genotypes (ICCV 92944 and JG 14) and heat sensitive genotype (ICC 10685) were identified
- Heat tolerant genotype JG 14 is characterized by early flowering, vigorous root biomass, large seed size fast grain filling rate and profuse nodulating type and has a tendency to avoid terminal heat,
- Chlorophyll fluorescence image changes when leaf is exposed to detrimental temperature, and significant genotypic variation was observed in chickpea. Therefore, changes in the fluorescence at high temperature were selected as the basis of thermo-tolerance.
- In lentil, it was observed that higher crop vigor, MSI, canopy temperature, high pollen viability, pod formation in terminal branches, pod development and grain filling rate are the most important parameters which was severely affected when temperature increased beyond 30°C. Twelve promising contrasting lines (IG 2507, IG 2543, IG 2598, IG 2821, IG 2559, IG 2960, IG3522, IG 4112, IG 4253, IG5128, WBL 58 and IG 4258) were identified based on Heat Susceptibility Index (HSI) and physiological parameters.
- Out of 26 wild germplasm lines of lentil screened under rainout shelter 4 genotypes (ILWL 432, ILWL236, ILWL 425 and ILWL 398) performed better in terms of crop establishment, NDVI, MSI, Biomass/plant, pods/plant and seed yield/plant.
- A unique RIL of lentil named as RILRT-200 was identified for early growth vigour based on plant height (25.0 cm) and NDVI (0.47) and early flowering (44 -52 days) and early maturity (95-100 days) compared to parental genotypes which can be utilized in lentil breeding program for developing the early maturing genotypes.
- Pigeon pea genotypes ICPL-20241, ICPL-87091, ICP-5028, MA-6, NDA-1, Bahar, IPA-203 and IPAC-211 showed tolerance against water logging stress whereas IPAC-202, RCM/DD-17, WB-2019/25, WB-2019/26, WB-2019/27 and IPAC-212 showed susceptibility against water logging stress. Tolerant group showed the formation of adventitious roots but susceptible group showed no adventitious roots.
- The Urdbean genotypes PGRU 95014 and PGRU 95016 were found to be putatively thermo tolerant and LBG 20, IPU 99-123 were found to be thermo sensitive using Temperature Induction Response (TIR) technique.

Biochemistry
A large number of pulse genotypes have been screened for a wide range of nutrients including carbohydrate, dietary fibre, protein, vitamins and minerals as well as bioactive secondary metabolites,viz., enzyme inhibitors, lectins, phytates, oligosaccharides, phenolic compounds and saponin.
-
- The genetic variability for various nutritional and anti-nutritional traits in chickpea genotypes was studied. The protein and phenol content was higher in the desi types as compared to the kabuli types. The antioxidant activity (AOA) was also assayed. The desi type chickpea varieties as well as the wild accessions had high levels of polyphenolic compounds and exhibited high levels of antioxidant activity.The phytic acid content in Kabuli types was higher (9.05 mg/g) as compared to the desi types (8.61 mg/g) and wild accessions (8.0 mg/g). No significant variation in folic acid content was recorded in chickpea genotypes. In the desi genotypes the folic acid content ranged from 136.1-152.7 µg/100g, whereas, in the kabuli types it ranged from 128.4-148.6µg/100g.
- Significant variation in nutritional quality of Chickpea was noted for protein (20.9 to 27.4%), soluble sugar (3.3 to 5.0%) and fat (2.8 – 4.6%). The most predominant fatty acid was linoleic acid.
- In pigeonpea, variation in the protein content (18.4 – 22.1%), starch (51.6 to 58.9%), soluble sugar (4.3 to 5.2%), polyphenols (0.3 to 0.6%), fat (1.25 to 2.45%) was noted and linoleic acid was the most predominating unsaturated fatty acid (43.59% of total fatty acid).
- In mungbean genotypes,variation in protein (20.6 to 24.1%), starch (45.8 – 49.5%), soluble sugar (7.1 to 8.9%) and crude fiber content (2.9 to 4.3%) with low fat content (below 2.3%) was recorded, whereas, in urdbean it was 18.7 – 24.6% protein, 43.6 – 48.6% starch, 8.3 to 8.8% soluble sugar and 4.0 to 5.4% crude fiber with fat content below 1.45%.
- In lentil, the protein (20.6 to 28.6%), starch (46.5 to 52.7%), soluble sugar (3.5-5.3%). Polyphenols 0.3%, total lipid (0.7 to 0.9%) with saturated fatty acid as palmitic and linoleic as the most predominating unsaturated fatty acid.
- Fieldpea contains 16.3 – 23.0% protein, 41.3 to 51.2% starch, 11.7 to 13.92% soluble sugar, 3.4 to 4.9% crude fiber with fat content of 1.1 to 1.5%. It contains palmitic acid as main saturated fatty acid whereas linoleic acid was the predominant unsaturated fatty acid.
- In Rajmash, protein (16.1 – 21.8%), starch (46.4 to 51.6%), soluble sugar (3.2 to 4.5%), crude fiber (3.9 to 5.7%) with fat content (2.2 to 5.0%) was recorded. Linoleic acid was the main unsaturated fatty acid.
- Lathyrus genotypes containing low neurotoxic compounds (BOAA) were identified. Such genotypes are Bio L – 208, Bio L 212 (Rattan) & Bio 202. These genotypes contain BOAA within safer limits. Dehulling and Pre – cooking /water soaking for 6 – 8 hrs reduced the BOAA content.
- Processing like dehulling, roasting/puffing, soaking and sprouting of pulses improve the nutritive value, digestibility of protein and availability of amino acids and minerals etc. by metabolizing anti-nutrients like polyphenols, protease inhibitors and oligosaccharides.
- Saponins of pulses help in lowering cholesterol, protect against cardiac diseases and cancer especially colon cancer, and protective against liver injury. Chickpea contain 0.21 – 0.35 and 0.41 – 0.57 mg/g sapogenol A and sapogenol B in grain of different genotypes. Soaking, germination and cooking reduce sapoganol A and B significantly. Pressure cooking almost destroys Sapogenol B content of grain.
- Pigeonpea contain 0.40 – 0.47 and 0.67 – 0.73 mg/g sapogenol A and sapogenol B in grain of different genotypes. Dehusking increases sapogenol A and B in grain, whereas cooking of dehusked grain significantly reduces sapogenol A and B.
- The fieldpea genotypes (EC 328758 and IPFD 2014-2) were found to have the highest antioxidant activity. Likewise, the cowpea varieties (GC 901 and PL3) were found to have the maximum antioxidant activity. The cowpea genotypes have much higher antioxidant activity than fieldpea genotypes.
- Cowpeas have a more diverse phenolic profile than field peas.
- Vegetable-type peas have very high amylose content (up to 85% of the total starch) and can be utilized to develop low glycemic index food.
- Verbascose and stachyose were identified as major RFOs in field pea.
- Stachyose and raffinose were found to be the major RFOs in common bean genotypes whereas verbascose was not detected in any of the investigated genotypes.
- Out of fifty common bean genotypes, three (Uday, EC 932147, and EC 931772) were found to have low stachyose content (<3%) and four (EC 932197, EC 931772, EC100098, and EC931866) were found to have low raffinose content (< 0.5 %).
- The total RFO content of common bean genotypes ranged from 3.2% (EC 931772) to 6.6% (EC 931861). Overall, clustering analysis identified six common bean genotypes having low total RFO content of 3.2 to 3.8 % (Uday, EC 932147, IC 360831, EC 931844, EC 931874, and EC 931772) with the least value noted for EC 931772 (3.2%).
Biochemical mechanism of heat tolerance
- Antioxidant defense mechanisms were higher in the heat-tolerant chickpea ICCV 92944 genotype as compared to heat-sensitive line ICC 10685. The tolerant lines showed higher activities of peroxidase and superoxide dismutase.
- The production of hydrogen peroxide and superoxide radicals was found to increase under heat stress as compared to normal conditions and it was observed that the increase in hydrogen peroxide and superoxide radical production was more in susceptible genotypes as compared to tolerant genotypes.
- It was observed that in chickpeas, four isozymes of SOD were present in the heat-tolerant genotypes whereas only three isozymes were present in the heat susceptible genotypes.
Microbiology
-
- BNF genetics in chickpea is being studied and twenty-six nodulation specific genes are identified following a homology-based search using CDC frontier genomic information as a subject and Medicago and soybean nodulation genes information as a query. Genes responsible for nodule initiation and development are localized in all 8 chromosomes of chickpea.
- SNPs and InDels in genes determining nodulation and symbiotic efficiency in chickpea were mined from 285 chickpea minicore genotypes following GATK tool and identified 2915827 bi-allelic SNPs and 6676215 InDels.
- Locus LOC101490264 is an ortholog of Medicago truncatula cytokinin response regulator 9 and has SNP in the exonic region which causes creation of stop codon in ICC1230 and ICC1710 genotypes and produces non-functional protein product. It’s well corresponding with nodule phenotypic data where these genotypes are low nodulator. Start lost variant caused by InDel is observed in the locus LOC101500280 of ICC20183 genotype which encodes Medicago truncatula ortholog VAPYRIN. This genotype is also a poor nodulator.
- Rajmash germplasm lines via IC417352, EC150250, IC356018 and IC356072 were found to be high nodulators. Nodule number varied from 3-15/plant. Nodule specific weight varied from 2-16mg/nodule.
- A series of experiments were conducted to optimize sampling time and interval for maximum extraction of root exudates under hydroponics system and Gibson tube. Root growth was comparatively better in conical flasks with aeration than the Gibson tubes. Based on the data, 6 to 12 days after inoculation is the suitable time for extraction of root exudates for trapping the signaling molecules. Current protocol is being standardized to improve induction of root nodules and extraction and analysis of signal molecules in root exudates.

Fig. Optimization of Hydroponics system for nodule induction in rajmash
-
- To break the yield barrier in chickpea, we are utilizing CRISPR/cas9 to knockout CKX gene coding for Cytokinin dehydrogenase. Cytokinin dehydrogenase plays a key role on metabolic degradation of cytokinin. Genome-wide analysis indicated the presence of 10 CKX genes in chickpea genome. Similarly, CaNCR genes from chickpea with high level nodule-tissue specific expression are selected for developing knockout mutants using CRISPR Cas9 tool.
- Nodule-specific cysteine rich (NCR) peptides of plant origin mediate terminal bacteroid differentiation in IRLC legumes. During the process of nodule development, free living rhizobial cells are differentiated into different morphotypes (E-Elongated/S-Spherical/U-Unmodified) of nitrogen fixing bacteroids.
- Sixty four IIPR CaNCR peptides including 16 novel peptides were identified in chickpea. Similarly, 32 CrNCR peptides including 8 novel peptides were identified in C. reticulatum. The predicted isoelectric point (Expasy pI) of Cicer-NCR peptides ranged between 3.57 and 9.65. Maximum number of NCR peptides possess pI in the range of 6.5-7.0 and 8.0-8.5. Hydropathicity in terms of GRAVY (Grand average of hydropathy) ranged between -1.137(CaNCR17) to +0.922 (CrNCR31). 34 out of 96 Cicer-NCR peptides possess positive GRAVY values.

Microbial Products With Commercial Potential
-
- Microbial consortium to mitigate moisture deficit stress
- Host-Specific potential Rhizobium strains:
| Crop | Rhizobium Strains | |
|---|---|---|
| Chickpea | RFC-119 |
 |
| Pigeonpea | IIPR-PR-315 | |
| Mungbean | IIPR-MR-127 | |
| Urdbean | IIPR-UR-32 | |
| Fieldpea | IIPR-FR-38 | |
| Lentil | LR2959 |
Agricultural Chemistry
Allelochemicals of Sorghum
-
- Chemical nature: Instrumental analysis (FTIR, 1HNMR, 13CNMR) of purified compounds of sorghum revealed them to be the derivatives of 3,4-dehydroxy anthocynadenes. Though all the extracted compounds of different solubility i.e. non-polar to polar contained identical flavanol skeletons as major chemical moieties in their chemical structures, however, they differ to each other in respect of functional substituents attached to flavanol skeleton at places R1, R2, R3 and R4.
- Bioefficacy of Allelochemicals: The allelo-compounds of sorghum were found active against seven soil borne fungus viz. Fusariumudum, Fusariumoxysporium sp. Ciceri, Rhizoctoniabatiticola, SclerotiniasclerotiorumAlternariaalternataSclerotiumrolfsii and Choenephoracucurbitarum of pulse crops. The LC50 values of isolated compounds against these fungus ranged from 19.0-130 µg ml-1.
- Activity against root knot nematodes: The Non-polar compounds of sorghum were found lethal to root knot nematodes. The emulsion concentrate formulation of non-polar allelo-compounds was found toxic to the juveniles at 300-700 g ml-1 at 24 and 48 hours of incubation. At 250 g ml-1it retarded hatching of test nematode up to the extent of 98%.
- Activity against weeds:
Formulation developed from allelo-chemicals of sorghum showed some adverse effect on vegetative growth of different winter weeds viz., Chenopodium album, Anagalisarvensis, Spergulaarvensis, Vicia sativa and Melilotusalba. The formulation from non-polar fraction of sorghum caused adverse impact on the development of purple nutsedge at doses ranging from 150-400 µg g-1 of soil.
Allelochemicals Of Sesame
-
- Activity against Cyprus rotundas: A considerable impact of emulsion concentrate formulation prepared from isolated mixtures of sesame root exudates was observed on the germination of purple nutsedge. EW formulation not only severely inhibited the germination but also caused considerable delay in germination of purple nut sedge.
- As growth retardant: The six formulations developed from 95% purified allelo fractions named as F, E, D, C, B and A of different solubility ranging from highly non-polar to completely polar nature of sesame root exudates was found toxic. Formulations of non-polar fractions, viz hexane (F) and ethyl acetate (E) were found extremely toxic at very low doses of chemicals ranging from 15-90 μg/g of soil. Compared to control, these two non-polar fraction formulations caused up to 90% reduction in total biomass of plants at their highest concentration (90 µg/g of soil).
- As inhibitor of root and tuber development: Emulsifiable concentrate formulations from highly non-polar (F) and non-polar (E) fractions, revealed a drastic reduction in total biomass of roots (including tubers) and in total number of newly developed tubers on roots. Compared to control 80-90% reduction in total root biomass and up to 95-99% reduction in tuber development tendency of roots was noticed at highest concentration (90µg/g of soil) of both of the formulations.
- Activity against other winter weeds of pulse crops: Apart from purple nutsedge, allelocompounds of sesame were also observed toxic to several other winter weeds, which generally infest the pulse crops. Emulsion concentrate or emulsive water (EW) formulation developed from crystallized product of sesame root exudates showed adverse effect on the germination and growth of prominent weeds. EW formulation at 240 g/g not only delayed but also inhibited germination of lambsquarters (Chenopodium album, L.) up to the extent of 80%, followed by scarlet pimpernel (Anagalisarvensis, L.) 75%, white sweet clover (Melilotus alba, L. Medik.) 65%, corn flurry (Spergulaarvensis, L.) 60%, fumitory (Fumariaparviflora, Lam.) 55% and common vetch (Vicia sativa, L.) 50% over control.
Herbicide Residue analysis
- The dissipation behavior and residue status of imazethapyr in soil and harvested urdbean grains were studied under field condition. More than 90% of imazethapyr was dissipated within 60 days after application in soils, with half-lives ranging in between 15.12 to 18.02 days. The final residues of imazethapy in harvested urdbean grains were below the LOQ.
- Dissipation behavior of glyphosate and aminomethyl phosphonic acid in soil and its residual effect on subsequent urdbean crop was studied. Glyphosate persisted in soil for up to 45 and 60 days, at application rate of 1.5 kg a.i./ha and 2.25 kg a.i/ha, respectively. Residues of glyphosate and AMPA were not detected in leaves and grains of subsequent urdbean crops at the time of harvest.
Protein content of chickpea varieties in India
Chickpea is a highly nutritious pulse crop that is dense in protein, fiber, and essential nutrients. It has numerous culinary uses, and incorporating them into our diet can help us meet our daily protein requirement and promote overall health. In this study, 47 chickpea varieties were evaluated for their total seed protein content and classified into different categories. Total protein content was determined using the Kjeldahl method with six replicates for all analyses as per the standard protocol (AOAC, 2005).


Results
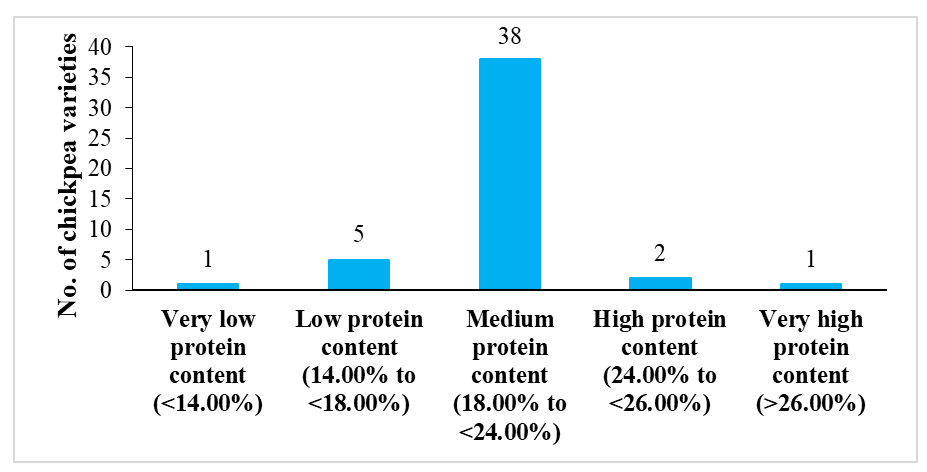
The chickpea varieties have a total seed protein content ranging from 13.36% to 26.67%. These varieties have been classified into five categories based on their total protein content:
- Very low protein content (<14.00%): Super Annigeri 1 (13.36% protein).
- Low protein content (14.00% to <18.00%): Phule G 0405 (15.94%), GBM-2 (16.38%), Indira Chana 1 (16.38%), JG 16 (16.93%), and RG 2015-08 (17.49%).
- Medium protein content (18.00% to <24.00%): 38 out of 47 varieties were found to have medium protein content.
- High protein content (24.00% to <26.00%): RSG 931 (24.00%) and CSJ 515 (24.39%).
- Very high protein content (>26.00%): T39-1 (26.67%).
Conclusion
The majority of the analyzed chickpea varieties are classified under the medium protein content category, ranging from 18.00% to less than 24.00%. Consequently, our institute is actively engaged in developing high-protein content chickpea varieties with a seed protein content exceeding 24.00%. This initiative aims to enhance the nutritional value of chickpeas, which, in turn, can have a positive impact on the public health and food industry.
| Sl. No. | Chickpea varieties | Protein content (%) | Sl. No. | Chickpea varieties | Protein content (%) |
|---|---|---|---|---|---|
| 1 | Super Annigeri 1 | 13.36 | 25 | IPC-2006-77 | 21.66 |
| 2 | Phule G 0405 | 15.94 | 26 | JSC 56 (RVG 203) | 21.85 |
| 3 | GBM-2 | 16.38 | 27 | NBeG 857 | 21.88 |
| 4 | Indira Chana 1 | 16.38 | 28 | BG 3062 | 21.91 |
| 5 | JG 16 | 16.93 | 29 | RG 2016-134 | 21.99 |
| 6 | RG 2015-08 | 17.49 | 30 | H 12-55 | 22.10 |
| 7 | Vijay | 19.52 | 31 | CSG 8962 | 22.28 |
| 8 | JG 2016-24 | 19.54 | 32 | BG 372 | 22.30 |
| 9 | GNG 2207 | 19.85 | 33 | NBeG 47 | 22.34 |
| 10 | KPG 59 | 19.96 | 34 | GL13001 | 22.38 |
| 11 | GNG 1581 | 20.03 | 35 | DCP 92-3 | 22.46 |
| 12 | GNG 2171 | 20.17 | 36 | Jaki 9218 | 22.51 |
| 13 | Pusa Chickpea 10216 | 20.44 | 37 | IPCL 4-14 | 22.53 |
| 14 | RSG 963 | 20.45 | 38 | BG 3043 | 22.70 |
| 15 | RVG 204 | 20.64 | 39 | JSC-55 (RVG 202) | 22.71 |
| 16 | RSG 888 | 20.70 | 40 | GCP 105 | 22.98 |
| 17 | GNG 1958 | 20.80 | 41 | GNG 2144 | 23.03 |
| 18 | BGM 20211 | 20.80 | 42 | Annigeri | 23.26 |
| 19 | BG 4005 | 20.86 | 43 | JG-11 | 23.32 |
| 20 | IPCMB 19-3 | 20.92 | 44 | Pant Gram 5 | 23.99 |
| 21 | Phule G 08108 | 21.17 | 45 | RSG 931 | 24.00 |
| 22 | JG 315 | 21.26 | 46 | CSJ 515 | 24.39 |
| 23 | AKG 1303 | 21.45 | 47 | T 39-1 | 26.67 |
| 24 | GNG 2299 | 21.64 |
Protein content of pigeonpea varieties in India
Pigeonpea is a nutrient-dense legume that is widely cultivated across the world. It is a rich source of protein, essential amino acids, vitamins, and minerals, making it a valuable addition to any diet. In this study, 23 pigeonpea varieties were evaluated for their protein content, and classified into different categories. Total protein content was determined using the Kjeldahl method with three replicates for all analyses as per the standard protocol (AOAC, 2005).


Results
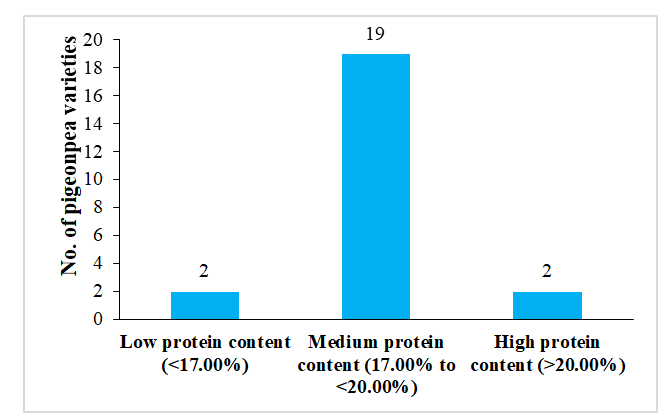
The total seed protein content in the pigeonpea varieties ranged from 16.25% to 20.67%. These varieties have been classified into three categories based on their total protein content.
- Low protein content (<17.00%): GRG-811 (16.25%), and WRGE93 (16.61%).
- Medium protein content (17.00% to 20.00%): 19 out of 23 varieties were found to have medium protein content.
- High protein content (>20.00%): Two varieties, i.e. Pusa Arhar-16 (20.02%) and IPH 15-03 (20.67%), showed more than 20 % protein.
Conclusion
Most of the pigeonpea varieties that were analyzed have a protein content of less than 20.00%. Only two varieties were found to have a protein content of over 20.00%. Therefore, there is a need to develop pigeonpea varieties that have a higher protein content. This will further enhance the nutritive value of pigeonpea.
| Sl. No. | Pigeonpea varieties | Protein content (%) | Sl. No. | Pigeonpea varieties | Protein content (%) |
|---|---|---|---|---|---|
| 1 | GRG-811 | 16.25 | 13 | BDN-716 | 18.69 |
| 2 | WRGE93 | 16.61 | 14 | PAU-881 | 18.69 |
| 3 | ICPL87119 | 17.06 | 15 | BLA-1 | 18.69 |
| 4 | Rajendra Arhar-2 | 17.33 | 16 | CORG-9701 | 18.69 |
| 5 | LRG-133-33 | 17.67 | 17 | PT-0012 | 18.71 |
| 6 | PT-0723-1-2-3 | >17.77 | 18 | Co-9 | 18.88 |
| 7 | TS3R | 17.79 | 19 | IPA-203 | 19.40 |
| 8 | UPAS 120 | 18.17 | 20 | JKM-189 | 19.44 |
| 9 | BDN-711 | 18.27 | 21 | IPA15-2 | 19.54 |
| 10 | IPH-9-5 | 18.36 | 22 | Pusa Arhar-16 | 20.02 |
| 11 | IPA 15-6 | 18.38 | 23 | IPH 15-03 | 20.67 |
| 12 | Pusa-992 | 18.54 |
Major Infrastructure/ Facilities

Instrument- Centrifuge

Instrument- Spectrophotometer

Instrument- Kjeldahl apparatus

Instrument- High-Performance Liquid Chromatography (HPLC) with PDA, RI, and Fluorescence detectors

Instrument- Chlorophyll fluorescence imaging

Instrument- Plant growth chamber

Instrument- Plant canopy analyzer

Instrument- Geen seeker

Instrument- Rainout shelter

Instrument- Dugout plot

Instrument- Green house
Scientists and their area of specialization















